Recombinant Antibody Production Service (monoclonal antibody, bispecific, nanobody/VHH expression)
LIBRA AbPro™-Recombinant Antibody Production Service
Recombinant Antibody Production Service
Based on the LIBRA™ antibody engineering platform, GeneMedi now provides antibody production services. In cooperation with the GM-LIBRA antibody expression group to achieve your antibody entities with high-throughout, high-quality, scalable, and state-of-the-Art Manufacturing.
Recombinant antibodies can be produced in various formats, including full-length, scFv, Fab, sdAb/VHH, Fc fusion protein, chimeric, and bispecific antibodies.
For more information, please contact us Inquiry.
LIBRA HuEasy™ is a Machine learning-based antibody humanization technology to balance high affinity with low immunogenicity. LIBRA HuEasy™ is an easier, faster and highly accurate, which reduce the wet lab validation procedure. For more information, please visit GeneMedi-LIBRA HuEasy™ website https://www.genemedi.com/i/BGMX-libra-hueasy-service.
LIBRA™ -Antibody Engineering Platform is GeneMedi’s innovative antibody engineering platform. LIBRA is developed with the characteristics such as Light, Intelligent, Balanced, Rational, and Algorithm-driven. LIBRA,(♎) is also Latin, meaning “Balance” in astrology. For more information, please visit GeneMedi-LIBRA™ website https://www.genemedi.com/i/about-LIBRA.
LIBRA AbProTM is GeneMedi’s professional antibody production platform for various antibody and antibody-conjugate formats. For more information, please visit GeneMedi-LIBRA AbPro™ website https://www.genemedi.com/i/BGMX-libra-abproduction-service.
Definition: What is Recombinant Antibody Production?
Recombinant antibodies (rAbs) are monoclonal antibodies which are generated in vitro using antibody coding genes without immunizing any animals or cultivating any hybridomas. This technology involves recovering antibody genes from source cells, amplifying and cloning the genes into an appropriate phage vector, introducing the vector into a host (bacteria, yeast, or mammalian cell lines), and achieving expression of adequate amounts of functional antibody. Recombinant antibodies maintain high standard of specificity and low immunogenicity. Recombinant antibodies are becoming indispensable tools for basic research, diagnostics, and clinical applications.
Recombinant antibodies can be produced in various formats: full-length, scFv, Fab, sdAb/VHH, Fc fusion protein, chimeric, and bispecific antibodies
Every full-length immunoglobulin (IgG) molecule consists of two light and heavy chains connected by disulfide bonds. Recombinant antibodies can come in different formats, including: full-length immunoglobulins (Ig), monovalent antibody fragments such as single-chain fragment variable (scFv) and fragment antigen-binding (Fab), and multimeric formats such as diabodies (dimeric scFvs) or triabodies (trimeric scFvs). Antibody fragments, like scFv, Fab and VHH, are small in size, thereby offering improved penetration of tissues or tumors compared to their full-length counterparts.
How to produce Recombinant Antibody?--Expression platform & Procedure & Protocol
The production of recombinant antibodies follows principally similar workflow. It consists of determining the sequence of the desired product followed by refinement of the codon, then gene synthesis and construct generation. Once the construct is delivered to the laboratory, expression constructs are produced, then they are transferred to a cell culture in the process called transfection and once the cell culture produces the desired recombinant antibody, it is regularly collected, purified and analyzed or used for further experimentation. For recombinant antibody production the stable cell lines such as CHO and HEK293 are used.
Case study: Data of Antibody Production
1. SDS-PAGE
The result of SDS-PAGE under reducing conditions shows that the purity of the antibody is >95%.

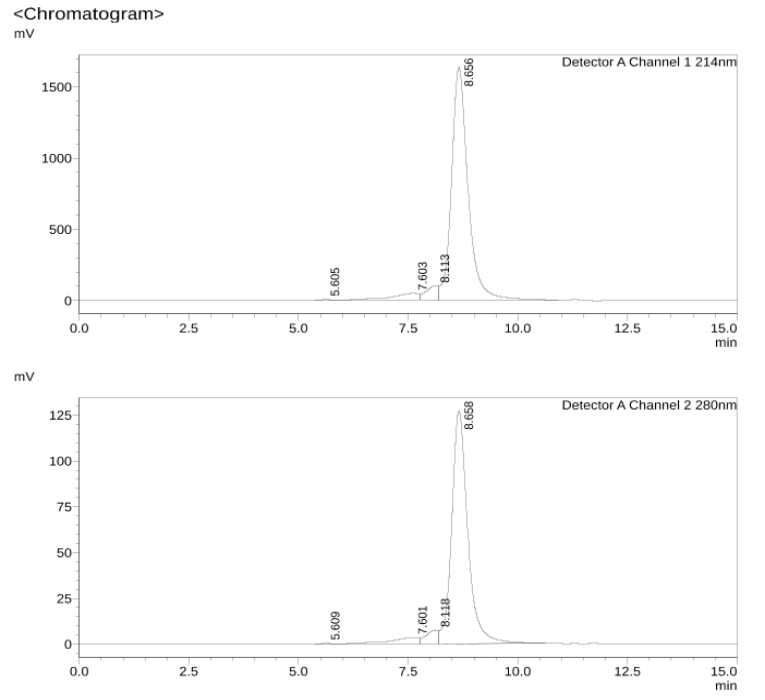
2. SEC-HPLC
The result of SEC-HPLC shows that the purity of the antibody is >90%.
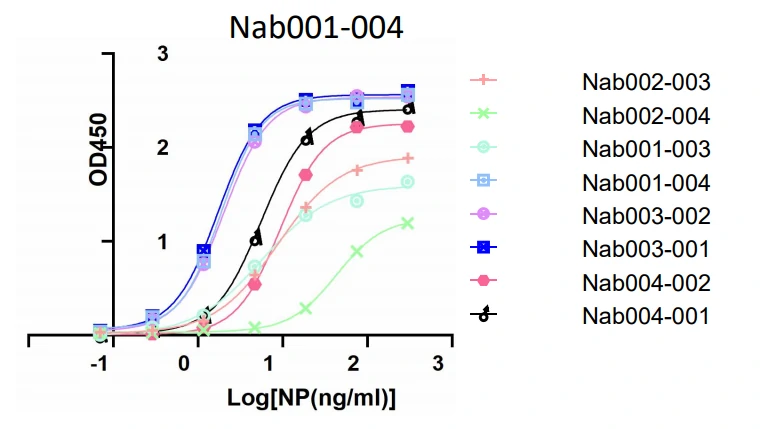
3. ELISA
GeneMedi’s SARS-CoV-2 NP Antibody Pairs Validation In Sandwich ELISA.